June 6, 2017- The U.S. Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC), are assisting state and local officials in assessing the risk of hepatitis A virus exposure from contaminated frozen tuna sourced from Sustainable Seafood.

Company, Vietnam, and Santa Cruz Seafood Inc., Philippines. If unvaccinated consumers have consumed the recalled product within the last two weeks, post-exposure prophylaxis may help prevent hepatitis A virus infection.
On May 18, Hilo Fish Company began recalling tuna sourced from Sustainable Seafood Company and Santa Cruz Seafood, Inc. that tested positive for the hepatitis A virus.
While the CDC is not currently aware of any illnesses linked to these products, it is advising post exposure prophylaxis (PEP) for unvaccinated persons who may have consumed the potentially contaminated tuna within the past two weeks.
The FDA is providing a list of establishments in TX, OK, and CA that may currently have potentially contaminated tuna in commerce to help alert consumers that may be at risk of the hepatitis A virus. Contact your health care professional if you believe you have been exposed to contaminated tuna.
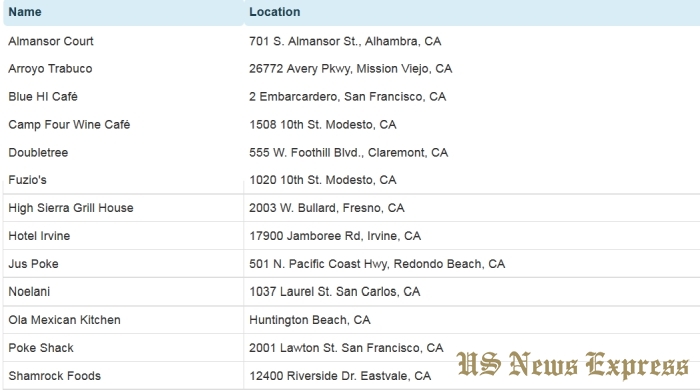
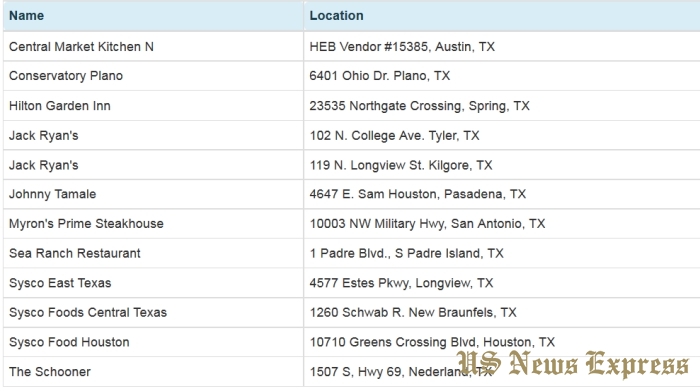
The current recall resulted from follow-up after the Hawaii Department of Health notified the FDA of a frozen tuna sample, sourced from PT Deho Canning Co.,which tested positive for hepatitis A on May 1, 2017. The initially recalled product has been removed from circulation and the newly recalled frozen tuna lots were not shipped to Hawaii, but were shipped to the mainland U.S.
Prior to FDA’s announcement, Hilo Fish Company alerted its customers and distribution partners directly to let them know about the company’s voluntary recall of certain tuna products on May 18. The FDA received records from the company or its distribution partners indicating that they sold frozen tuna to the establishments listed on the FDA’s website. The FDA is working with Hilo and other distributors to ensure that the companies remove product from the market. The table containing the names of establishments have been updated.

It is the responsibility of the Hilo Fish Company to notify its customers about the voluntary recall. It is also the responsibility of any company that received a recall notice from Hilo Fish Company to notify its customers. The establishments identified on the FDA’s website should have received a notice from Hilo Fish Company or their direct supplier. If they have not, they should reach out to their suppliers for more information. Any company that has questions about the voluntary recall or has affected product and did not receive notice should contact the FDA at 1-800-SAFEFOOD.



















